 NAEOTOM Alpha is the world’s first photon counting Computer Tomograph
NAEOTOM Alpha is the world’s first photon counting Computer Tomograph- First new major technological improvement for Computed Tomography (CT) imaging in nearly a decade
- Revolutionary images provided by photon-counting CTs, more people all over the world will benefit from precise and comprehensive examinations at low radiation and contrast dose
Siemens Healthineers announces the Food and Drug Administration (FDA) 510 (k) clearance of the NAEOTOM Alpha, the world’s first photon counting computer tomograph. With a novel system concept and pioneering new detector technology, it ushers in a new era in computed tomography, a technology that determines the course of many medical decisions.
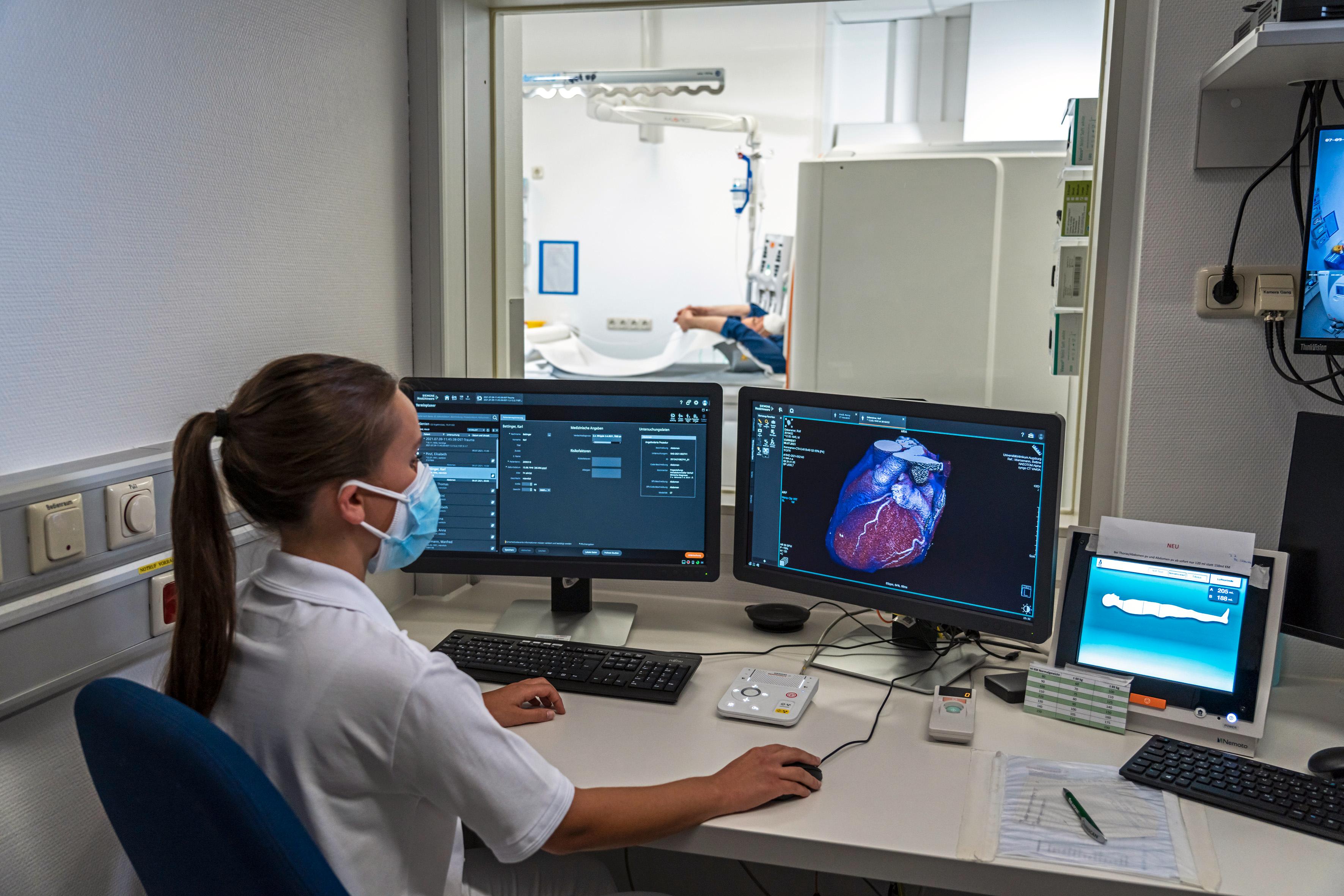
Thanks to the revolutionary images provided by photon-counting CTs, more people all over the world will benefit from precise and comprehensive examinations at low radiation and contrast dose – from oncological procedures and heart diagnostics to lung follow-up checks for respiratory illnesses. The centerpiece of this innovation is the new photon-counting detector whose active detection layer consists of a cadmium telluride one crystal (CdTe) and offers clear advantages over conventional CT detectors.
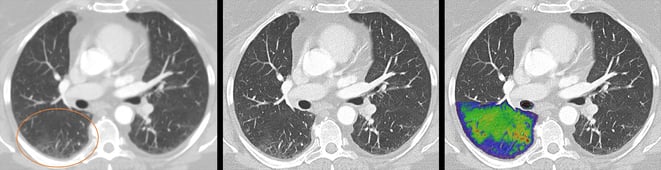
Standard CT detectors convert the X-rays in a two-step process first into visible light that is subsequently detected by a light sensor, ultimately producing the final image. Due to this intermediate step, important information about the energy of the X-rays is lost and no longer available to aid in diagnosis; contrast is reduced, and images lack clarity. The photon-counting CT detector developed by Siemens Healthineers no longer converts the X-rays into visible light. The X-ray photons are converted directly into completely digital electrical signals and then counted without information loss.
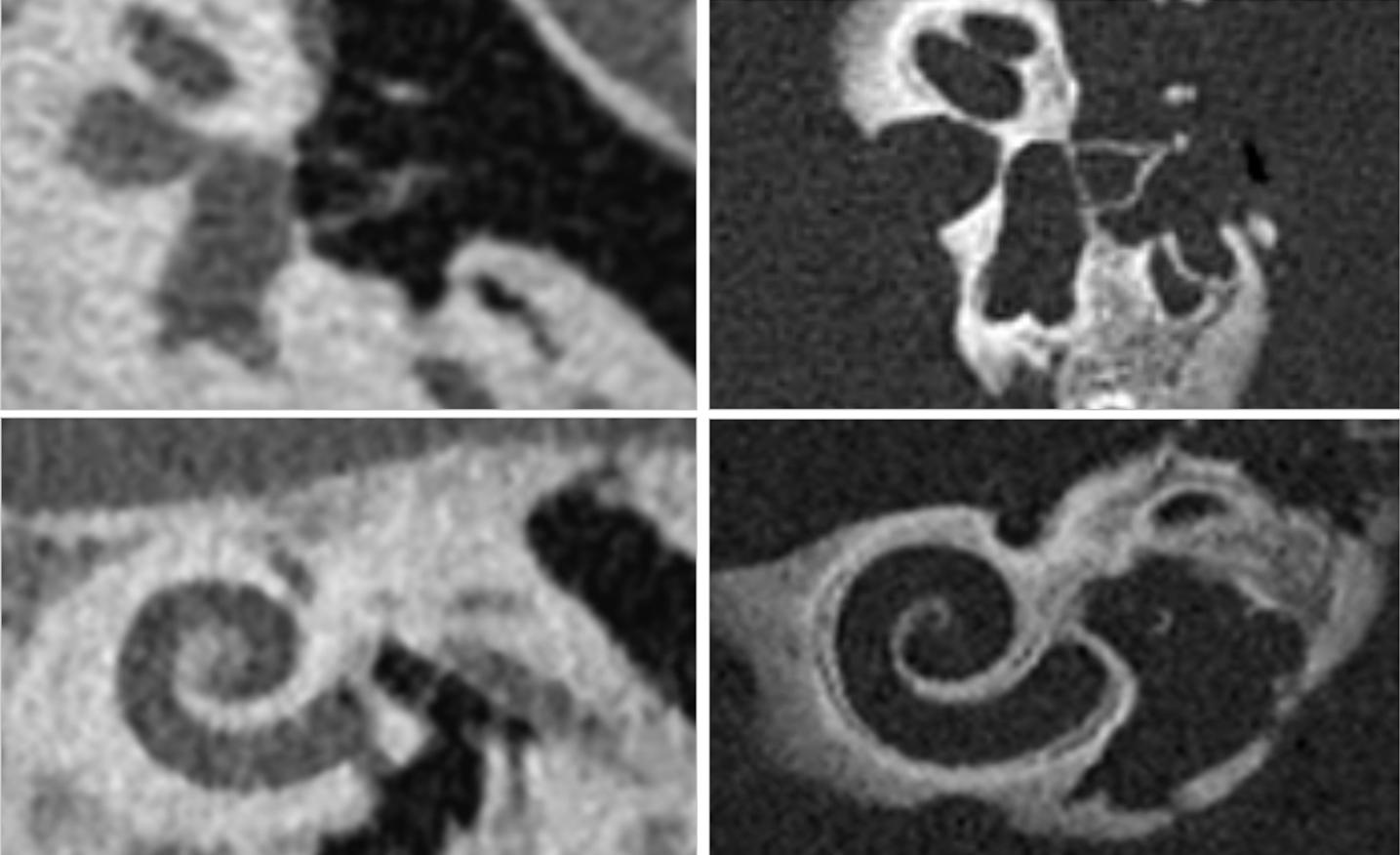
This adds a wealth of completely new clinically relevant information and improves image sharpness and contrast. “About 15 years ago, work on photon counting and its clinical vision started at Siemens Healthineers. We always believed in the tremendous clinical value and relentlessly worked on it together with our partners. We are excited that we have received FDA 510(k) clearance,” says André Hartung, Head of Diagnostic Imaging at Siemens Healthineers.
The company will have more to share about this leap on computed tomography at its Siemens Healthineers SHAPE 22 Event on November 15th. Three CT-experts from Siemens Healthineers were recently nominated for the German Future Award for the development of Photon-Counting-CT.

Siemens Healthineers Media Contact
Jeff Bell
Phone: (484) 868-8346; Email: jeffrey.t.bell@siemens-healthineers.com
Siemens Healthineers AG (listed in Frankfurt, Germany: SHL) is shaping the future of Healthcare. As a leading medical technology company headquartered in Erlangen, Germany, Siemens Healthineers enables healthcare providers worldwide through its regional companies to increase value by empowering them on their journey towards expanding precision medicine, transforming care delivery, improving the patient experience, and digitalizing healthcare.
Siemens Healthineers is continuously developing its product and service portfolio, with AI-supported applications and digital offerings that play an increasingly important role in the next generation of medical technology. These new applications will enhance the company’s foundation in in-vitro diagnostics, image-guided therapy, and in-vivo diagnostics. Siemens Healthineers also provides a range of services and solutions to enhance healthcare providers’ ability to provide high-quality, efficient care to patients.
In fiscal 2020, which ended on September 30, 2020, Siemens Healthineers, which has approximately 54,000 employees worldwide, generated revenue of €14.5 billion and adjusted EBIT of €2.2 billion. Further information is available at www.siemens-healthineers.com.
Meet the Author
Cassling strengthens community healthcare through customer-centric imaging and therapeutic technology, services and solutions. From critical access hospitals and diagnostic imaging centers to large health systems and IDNs, Cassling is committed to helping healthcare organizations improve access and outcomes, create efficiencies and lower costs.



Comments